The platinum group metals or platinoids are a collection of six silvery-white chemical elements (with the exception of osmium, which has a bluish-white tint), having similar physical and chemical properties and included in the list of precious (noble) metals. Among them are:
- Jewelry industry
- Iridium is a hard, refractory metal that has the highest level of corrosion resistance even at elevated temperatures.
- Osmium is the heaviest metal and is difficult to machine.
- Palladium is a silvery-white ductile metal found in nature in the form of crystals.
- Platinum is one of the rarest and most expensive among all noble metals.
- Rhodium is a hard metal mineral that reflects electromagnetic rays well in the visible image spectrum.
- Ruthenium is a refractory, highly inert metal.
Platinum group metals: list
The group consists of the following six chemical elements, arranged in order of increasing atomic weight:
- Ru – ruthenium.
- Rh – rhodium.
- Pd – palladium.
- Os – osmium.
- Ir – iridium.
- Pt – platinum.
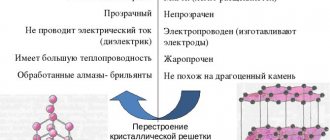
The platinum group metals are silvery-white in color, with the exception of osmium, which is bluish-white. Their chemical behavior is paradoxical in that they are highly resistant to most reagents, but are used as catalysts that easily accelerate or control the rate of oxidation, reduction, and hydrogenation reactions.
Ruthenium and osmium crystallize into a hexagonal close-packed system, while others have a face-centered cubic structure. This is reflected in the greater hardness of ruthenium and osmium.
Storage and transportation
The procedure for handling precious metals and stones, including platinoids, is strictly regulated by the relevant Instructions approved by the Ministry of Finance of Russia by Order No. 231n dated 12/09/16. According to which, the safety and protection of valuables from harmful influences must be ensured throughout all possible operations. The list of possible operations includes:
- appeal,
- production,
- processing,
- usage,
- transportation,
- storage.
Responsibility for fulfilling the requirements for the storage and transportation of precious metals lies with managers, financially responsible persons, as well as direct performers.
History of discovery
Although platinum-bearing gold artifacts date back to 700 BC. e., the presence of this metal is more likely an accident than a pattern. Jesuits in the 16th century mentioned gray dense pebbles associated with alluvial gold deposits. These stones could not be melted, but they formed an alloy with gold, and the ingots became brittle and could no longer be cleaned. The pebbles came to be called platina del Pinto - granules of silvery material from the Pinto River, which flows into the San Juan River in Colombia.
Malleable platinum, which can only be obtained after complete purification of the metal, was isolated by the French physicist Chabanot in 1789. It was used to make a cup presented to Pope Pius VI. The discovery of palladium in 1802 was reported by the English chemist William Wollaston, who named the chemical. element of the platinum group of metals in honor of the asteroid. Wollaston subsequently claimed to have discovered another substance present in platinum ore. He named it rhodium because of the pink color of the metal salts. The discoveries of iridium (named after the rainbow goddess Iris because of the variegated color of its salts) and osmium (from the Greek word for "smell" because of the chlorine odor of its volatile oxide) were made by the English chemist Smithson Tennant in 1803. French scientists Hippolyte-Victor Collet-Descoti, Antoine-François Fourcroy, and Nicolas-Louis Vauquelin isolated the two metals simultaneously. Ruthenium, the last element isolated and identified, received its name from the Latin name for Russia from the Russian chemist Karl Karlovich Klaus in 1844.
In contrast to substances such as gold, silver, which are easily isolated in a relatively pure state by simple fire refining, platinum group metals require complex water-chemical processing. These methods were not available until the late 19th century, so identification and isolation of the platinum group lagged behind silver and gold by thousands of years. Additionally, the high melting point of these metals limited their use until researchers in Britain, France, Germany, and Russia developed methods to convert platinum into a workable form. How platinum group precious metals have been used in jewelry since 1900. Although such applications remain relevant today, industrial applications have far surpassed them. Palladium became a highly sought after contact material in telephone relays and other wired communication systems, providing long life and high reliability, and platinum, because of its resistance to spark erosion, was used in combat aircraft spark plugs during World War II.
After the war, the expansion of molecular conversion techniques in petroleum refining created a huge demand for the catalytic properties possessed by platinum group metals. By the 1970s, consumption increased even further when automobile emissions standards in the United States and other countries led to the use of these chemicals in catalytic conversion of exhaust gases.
Ore
With the exception of small placer deposits of platinum, palladium and osmic iridium (an alloy of iridium and osmium), there is practically no ore in which the main component is the chemical element - a platinum group metal. The minerals are typically found in sulfide ores, particularly pentlandite (Ni, Fe)9S8. The most common are laurite RuS2, irarsite, (Ir,Ru,Rh,Pt)AsS, osmiridium (Ir,Os), cooperite, (PtS) and braggite (Pt,Pd)S.
The world's largest deposit of platinum group metals is the Bushveld complex in South Africa. Large reserves of raw materials are concentrated in the Sudbury deposits in Canada and the Norilsk-Talnakh deposits in Siberia. In the United States, the largest deposits of platinum group minerals are located in Stillwater, Montana, but there they are much smaller than in South Africa and Russia. The world's largest platinum producers are South Africa, Russia, Zimbabwe and Canada.
World reserves
At the beginning of the 3rd millennium, confirmed platinum deposits by country of the world are:
- South Africa - 23,950 tons.
- Russia – 2200 tons.
- Zimbabwe - 326 tons.
- Canada - 200 tons.
- China - 120 tons.
- Global reserves are 27,050 tons.
Reserves of other platinoids (iridium, osmium, palladium, rhodium, ruthenium):
- South Africa - 49,920 tons.
- Russia – 6,220 tons.
- Zimbabwe - 594 tons.
- Canada - 520 tons.
- USA - 310 tons.
- Global resources are estimated at 58,170 tons.
Mining and enrichment
The main South African and Canadian deposits are exploited by mining. Virtually all platinum group metals are recovered from copper or nickel sulfide minerals using flotation separation. Smelting the concentrate produces a mixture that is washed out of copper and nickel sulfides in an autoclave. The solid leach residue contains 15 to 20% platinum group metals.
Gravity separation is sometimes used prior to flotation. The result is a concentrate containing up to 50% platinum metals, eliminating the need for smelting.
Mechanical properties
Platinum group metals differ significantly in their mechanical properties. Platinum and palladium are quite soft and very malleable. These metals and their alloys can be worked in both hot and cold conditions. Rhodium is first worked hot and can later be worked cold with fairly frequent annealing. Iridium and ruthenium must be heated; they cannot be cold processed.
Osmium is the hardest of the group and has the highest melting point, but its tendency to oxidize has its limitations. Iridium is the most corrosion-resistant of the platinum metals, and rhodium is valued for maintaining its properties at high temperatures.
Platinum
Platinum has been known since ancient times, and the name comes from the Spanish language: “little silver.” The conquistadors were the first Europeans to become acquainted with platinum in the mid-16th century in South America. And in 1735, the king of Spain issued a decree according to which platinum should be drowned in the river and not brought into the country. Later the river was named Platino del Pinto. The decree was repealed only forty years later, for the falsification of gold and silver coins by the Madrid authorities. Later, alchemists became acquainted with platinum, but considered it unsuitable. But in France it still found its use in the form of a standard meter and then a kilogram. In Russia, the metal was called white gold, having been found in placer gold.
Platinum is one of the rarest metals, because its content in the earth's crust is low. The main deposits of platinum are: Russia, USA (2 large mines: Stillwater, East Bouder), China, Zimbabwe (9 tons), South Africa (Bushwell complex).
In Russia, this PGM metal has a unique base, with a huge number of deposits, the main of which is located in Zarechensk (Murmansk region).
Platinum mining is a labor-intensive process, of two types:
- Open mining method: in secondary placer deposits. There, platinum is concentrated as a result of the destruction of the primary rock.
- Closed mining method: in primary deposits, with the construction of mines, drilling holes in rocks, laying explosives and the explosion itself.
Platinum is a precious metal used in jewelry and industry. The properties of platinum are wide:
- heavier than gold and silver;
- does not oxidize;
- does not react with other elements when heated;
- ductile metal;
- has good electrical conductivity;
- does not dissolve under the influence of acids;
- high melting point;
- good malleability.
The scope of platinum is quite wide and is not limited to jewelry production, because it has unique beauty and nobility. Currently, the price of metal is growing, as it is also used in industry. So, the uses of platinum:
- jewelry production, for the manufacture of various types of jewelry;
- medicine;
- chemical industry;
- shipbuilding;
- aircraft manufacturing;
- space sphere;
- glass industry;
- banking.
Structural Applications
Because clean annealed platinum is so soft, it is susceptible to scratches and deterioration. To increase hardness, it is alloyed with many other elements. Platinum jewelry is very popular in Japan, where it is called "hakkin" and "white gold." Jewelry alloys contain 90% Pt and 10% Pd, which is easy to process and solder. The addition of ruthenium increases the hardness of the alloy while maintaining oxidation resistance. Alloys of platinum, palladium and copper are used in forged products because they are harder than platinum-palladium and less expensive.
Crucibles used to produce single crystals in the semiconductor industry require corrosion resistance and stability at high temperatures. Platinum, platinum-rhodium and iridium are best suited for this application. Platinum-rhodium alloys are used in the production of thermocouples, which are designed to measure elevated temperatures up to 1800 °C. Palladium is used both in pure and mixed form in electrical devices (50% of consumption) and in dental alloys (30%). Rhodium, ruthenium and osmium are rarely used in their pure form - they serve as an alloying additive for other platinum group metals.
Catalysts
About 42% of all platinum produced in the West is used as a catalyst. Of these, 90% are used in automotive exhaust systems, where refractory pellets or honeycomb structures coated with platinum (as well as palladium and rhodium) help convert unburned hydrocarbons, carbon monoxide and nitrogen oxides into water, carbon dioxide and nitrogen.
An alloy of platinum and 10% rhodium in the form of a red-hot metal mesh serves as a catalyst in the reaction between ammonia and air to produce nitrogen oxides and nitric acid. When fed with a mixture of ammonia, methane can produce hydrocyanic acid. In petroleum refining, platinum on the surface of alumina beads in a reactor acts as a catalyst to convert long-chain petroleum molecules into branched isoparaffins, which are desirable in high-octane gasoline blends.
Osmium
Pure osmium does not exist in nature; it is found in a complex with rhodium. Osmium deposits are concentrated in: Africa (south), Tasmania, Australia, USA, Canada, Russia, Colombia, South Africa (the largest reserves).
Characteristics of osmium:
- silver-blue crystals;
- pungent odor when interacting with substances;
- insoluble in alkalis and acids, mixtures of hydrochloric and nitric acids;
- chemical inertness;
- Powdered osmium is a raw material for products, slowly susceptible to dissolution;
- hard and brittle;
- refractory;
- high density: the heaviest metal.
Electroplating
All platinum group metals can be plated. Because of the hardness and shine of the resulting coating, rhodium is most often used. Although its cost is higher than platinum, its lower density allows it to use less material at a comparable thickness.
Palladium is a platinum group metal that is the easiest to use for coating. Thanks to this, the strength of the material increases significantly. Ruthenium has found use in tools designed for low-pressure friction machining.
Palladium
Today, palladium is the most expensive platinum group metal used in industry.
In the late 17th century, Brazilian mining workers found an alloy with similar gold or platinum content. The chemist W. Wollaston worked on the interaction of crude platinum with hydrochloric and nitric acids, and the addition of mercuric cyanide. Afterwards a yellow precipitate fell, which he heated with sulfur and borax. Having received balls of metal and calling the metal palladium.
The metal was named after the asteroid. This platinum group metal is found very rarely in nature in its pure form. And more often in combination with other metals (gold, silver, platinum).
Metal is also mined in two ways:
- on indigenous (primary) deposits;
- in placer form, in the form of a nugget, and account for only 2% of all metal production.
Main palladium deposits:
- Canada;
- USA;
- Colombia;
- Austria;
- SOUTH AFRICA.
Palladium mining also occurs in Russia, concentrated in the Urals. The main properties of palladium are:
- low density;
- chemical inertness;
- looks like silver;
- high malleability;
- elasticity, which allows it to be used in the manufacture of jewelry;
- melting point 1555 degrees;
- processable;
- fragile metal under mechanical stress;
- dissolves when exposed to a mixture of sulfuric and nitric acids;
- can form compounds with other chemical elements (boron, chlorine, sulfur, silicon);
- does not react;
- does not oxidize in nature.
Areas of application of palladium
- Jewelry sphere, adding metal to alloys;
- production of catalysts for cars, for afterburning exhaust gases according to EU standards;
- medicine: production of elements for pacemakers, instruments, utensils;
- chemical industry: manufacture of flasks and containers, as well as for hydrogen purification;
- electronic sphere: military aerospace technology;
- investment activity: purchase of bullion.
Chemical compounds
Organic platinum group metal complexes, such as alkylplatinum complexes, are used as catalysts in the polymerization of olefins, the production of polypropylene and polyethylene, and the oxidation of ethylene to acetaldehyde.
Platinum salts are increasingly used in cancer chemotherapy. For example, they are part of drugs such as Carboplatin and Cisplatin. Ruthenium oxide coated electrodes are used in the production of chlorine and sodium chlorate. Rhodium sulfate and phosphate are used in rhodium plating baths.